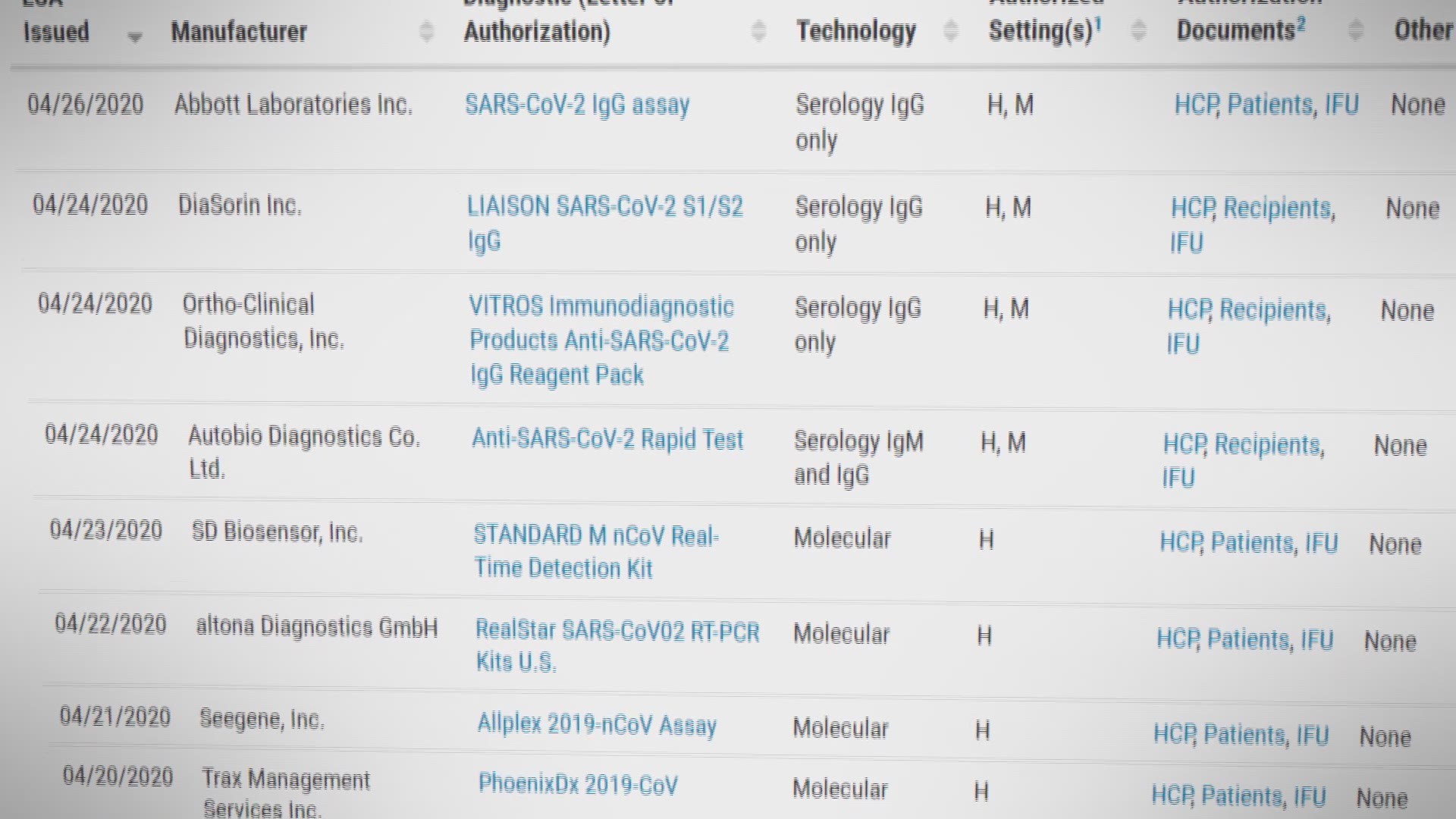
39 A biotinylated version of mAb HP6017, which binds to the Fc part of human IgG, was used as the detection antibody. Finally, sera of vaccinated individuals were tested with the new ELISAs before and after vaccination.įor the quantification of human IgG, well‐characterised monoclonal antibodies were used, which bind to all IgG subclasses with high specificity and affinity. For standardisation and to calculate conversion factors, international WHO references for immunoglobulins 36 and SARS‐CoV‐2‐specific antibodies 37 were used. The results of the study group using our newly developed ELISAs were compared to those of commercially available anti‐SARS‐CoV‐2 IgG immunoassays from EuroImmun, Lübeck, Germany and Thermo Fisher Scientific, Darmstadt, Germany. In order to validate the developed ELISAs, a study group of individuals with positive SARS‐CoV‐2 PCR test results (true positives) and a control group with sera taken before the COVID‐19 pandemic (true negatives) were used for ROC analysis. The measurement of antibodies against both immunodominant SARS‐CoV‐2 antigens usually allows for the differentiation of the immune response after vaccination and infection, as almost all licenced vaccines target the spike protein. The aim of this study was to develop quantitative enzyme‐linked immunosorbent assays (ELISA) in order to detect the binding of human IgG to the Spike‐S1 and nucleocapsid virus proteins. In the case of anti‐SARS‐CoV‐2 immunoglobulin, an international standard now exists 37 that has been evaluated in a WHO collaborative study. 35 Antigen‐specific IgG concentrations are determined using a calibration curve traceable to international human immunoglobulin standards, 36 thus enabling values to be compared across laboratories if the same antigens and assay components are used. 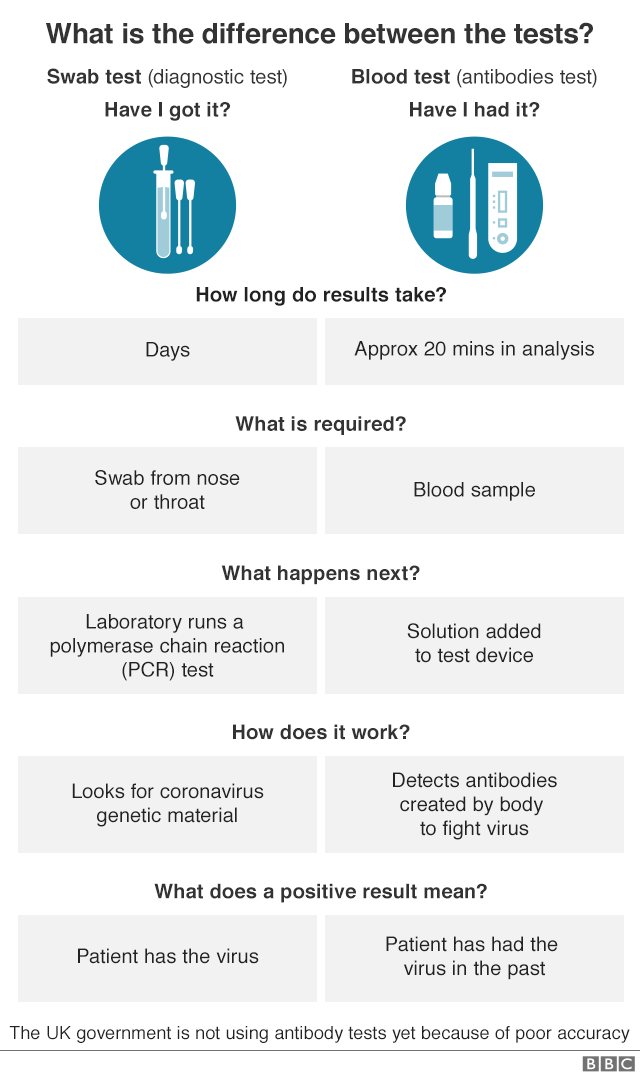
For example, the quantification of antigen‐specific human IgG antibodies is an important component in the diagnosis of hypersensitivity pneumonitis. Quantitative immunoassays are part of the standard repertoire in serology diagnostics and research.  
Neutralising antibody titres were found to be strongly associated with the magnitude of the IgG response 30, 31, 32, 33 and were highly predictive of the immune protection from symptomatic SARS‐CoV‐2 infection. On the one hand, a significant increase in antibody concentration indicates successful vaccination on the other hand, the antibody level may indicate the probability and duration of immune protection and sterilising immunity. 28Īs the vaccination campaign progresses and breakthrough infections occur even after full vaccination, 29 immunoassays for the quantitative detection of anti‐SARS‐CoV‐2 antibodies are becoming increasingly import. 26, 27 The long‐term persistence of SARS‐CoV‐2‐specific antibodies is unclear as is whether the presence of antibodies confers protective immunity against SARS‐CoV‐2. 24, 25 Specific IgG antibody titres decrease over time but remain elevated up to 6 months after a complete vaccination. After the first vaccination, antibody responses are elicited and then boosted with the second vaccine dose to protein‐binding IgG levels above those of human convalescent sera. 20 Because antibodies with proven neutralising activity bind to the spike protein and the RBD in particular, 21, 22, 23 the spike protein is also the major antigen target for vaccines. The S1 subunit contains the receptor‐binding region (RBD) and shows less homology to other coronaviruses than the S2 protein. The spike protein is located on the surface of the enveloped RNA virus and consists of trimers of two glycosylated subunits (S1 and S2). 17, 18 Normally, the nucleocapsid protein is present in cells at higher copy numbers than the spike protein 19 and is responsible for the high sensitivity of immunoassays based on this antigen due to its high immunogenicity. 12, 13, 14, 15, 16 Both the spike protein that binds to the receptors on the host cell for virus entry and the nucleocapsid protein required for virus replication are recognised by human antibodies and serve as antigens for the serological detection of infection. 8, 9 Numerous immunoassays have been developed to detect IgG to the SARS‐CoV‐2 antigens, 10, 11 some of which are characterised by high specificity and sensitivity. 1, 2, 3, 4, 5, 6, 7 IgG antibodies in particular remain detectable after the end of the disease or even after infections without symptoms. Specific detection of the immune response to SARS‐CoV‐2 infection plays an important role in tracking the spread of COVID‐19 in the population.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
